Major Corrosion Issues in Industrial Water Systems – Part 1
Brad Buecker, SAMCO Technologies
Posted 3/12/2026
Introduction – Major Corrosion Issues in Industrial Water Systems
Water is utilized for many cooling and process applications at a vast array of industries throughout the world. But water is also widely considered to be the nearest thing to a universal solvent, which means that it, and especially dissolved compounds such as acids, bases, oxygen, etc., can initiate corrosion of metals (and some non-metals) in almost any water network. In this series, we will examine some of the most important water system corrosion mechanisms, with discussion of preventive measures to minimize attack. Many books have been written about corrosion, and we will only scratch the surface (pardon the pun) in this series, but the key point is that for any system design, many factors need to be evaluated to select proper corrosion mitigation programs. An excellent resource for further information is the Association for Materials Protection and Performance (www.AMPP.org).
Part 1 examines important corrosion fundamentals. The information will provide a foundation for subsequent discussion in later installments.
Some Metallurgy Fundamentals
Mild carbon steel is the usual material for cooling water and service water system piping, heat exchanger shells, boiler tubes, and other applications due to its high strength at lower cost than other metals. Heat exchanger (HX) tubes or plates are often fabricated from stainless steel, copper alloys, titanium, aluminum, or perhaps in some specialty cases, expensive corrosion-resistant metals. Galvanized steel components are common in cooling towers, and smaller towers may predominantly be of galvanized construction.
Unit cells are the fundamental building blocks of typical metals, and Figure 1 shows the three most common unit cell structures.

Common water system metals usually have either the bcc or fcc unit cell structure. Important examples include:
- Mild carbon steel, which is the most widely used metal in industry, has a “bcc” structure from ambient to relatively high temperatures. Appendix A outlines the composition of the typical steels for steam generator applications and gives a brief outline of common alloying elements.
- The addition of nickel to steel induces formation of the “fcc” structure, which, at 8% or greater nickel content, exists at ambient temperature.
- Chromium above 12% concentration imparts the “stainless” aspect to stainless steels.
- The combination of the two properties above results in the austenitic stainless steels, with the most popular being the 304 and 316 varieties.
- Copper alloys typically have a “fcc” structure
A key issue that significantly influences metal properties and, at times, corrosion susceptibility is that of metal grains and grain boundaries. Virtually all metals utilized in conventional applications are not one single crystal, but “are composed of a collection of many crystals or grains.”1

Depending on the fabrication process and the application requirements, grains may range from small to large. Certain fabrication methods such as cold rolling can deform grain structure and impart stress in the metal, which in turn may influence corrosion susceptibility. Accordingly, many metals after initial fabrication go through various forms of heat treatment and annealing to relieve internal stress and establish desired grain properties.
Other important features or anomalies besides grain boundaries that can influence corrosion in metals include:2
- Missing atoms and voids
- Presence of foreign atoms
- Interstitial atoms. These are small atoms that fit within the metal atoms of unit cells. Carbon in low concentrations sits in interstitial locations.
- Surface roughness
- Metallurgical transformations induced by procedures such as welding.
Corrosion Basics
Metallic corrosion is an electrochemical mechanism, although in some cases such as erosion-corrosion and cavitation, mechanical influences play a part. The concept of anodes and cathodes is fundamental for understanding corrosion processes. Consider the example of a carbon steel coupon immersed in an acidic solution, in this case dilute hydrochloric acid (HCl).

Metal loss (“oxidation”) occurs at anodes, releasing ferrous ions (Fe2+) into solution. The free electrons flow through the metal to cathodes, where they react with hydrogen ions from the acid (“reduction”) to produce hydrogen gas (H2) that leaves the chloride ions behind. The basic equations are:

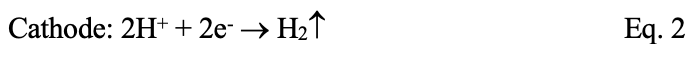


The following table highlights half-cell potentials for several of the most common metals as compared to the standard hydrogen half-cell.

From this table, we can derive several important concepts.
- The metals above hydrogen in the table will all normally corrode in acid solutions.
- Copper (and typically its alloys) are on the “noble” side of the hydrogen half-cell potential, meaning that it remains stable in common mineral acids. Oxidizing agents are usually needed for corrosion.
- Silver and gold are two truly noble metals. Especially for gold, special solutions, e.g., aqua regia, are needed to dissolve the metal.
- This data is also valuable for determining the probability of galvanic corrosion when two dissimilar metals are coupled in a water environment. Galvanic corrosion will be discussed in a later installment of this series.
Other cathodic reactions are possible, with by far the most common being oxygen reduction in neutral or alkaline waters, for example the circulating water in cooling tower-supplied circuits.
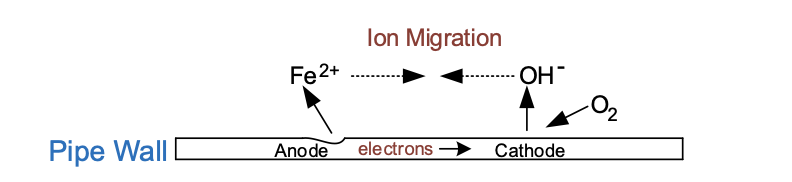
The anodic reaction is the same as previously shown.

Oxygen is reduced at the cathode:

The hydroxide ions combine with Fe2+ to form ferrous hydroxide:

In the oxygen-laden environment, additional reactions occur. Ferrous hydroxide will continue reacting with oxygen to form ferric hydroxide:
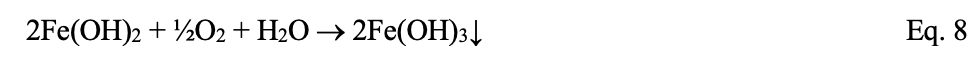
Besides the corrosion induced by oxygen attack, the reaction products convert to rust (Fe2O3∙xH2O) that can accumulate in pipes and restrict flow.
Two other important aspects to consider are that in the electrochemical process, cathodic reactions dictate the rate of corrosion, while anodic reactions dictate the type of corrosion. As will be further outlined, situations can arise with a small number of fixed anodes in a large cathodic environment. This combination can lead to severe localized corrosion and potentially rapid failures.
An important point to note is that the half-cell potential for the oxygen reaction shown in Figure 4 and highlighted in Equation 6 is -0.401V in neutral and alkaline solutions.2 Thus, oxygen will react with some metals that are noble to acid. Most notable are copper and copper alloys, where dissolved oxygen combined with a complexing agent such as ammonia can be quite corrosive. Examples will appear in later parts of this series.
A number of other factors can influence most corrosion mechanisms. Several of the most important are illustrated below.
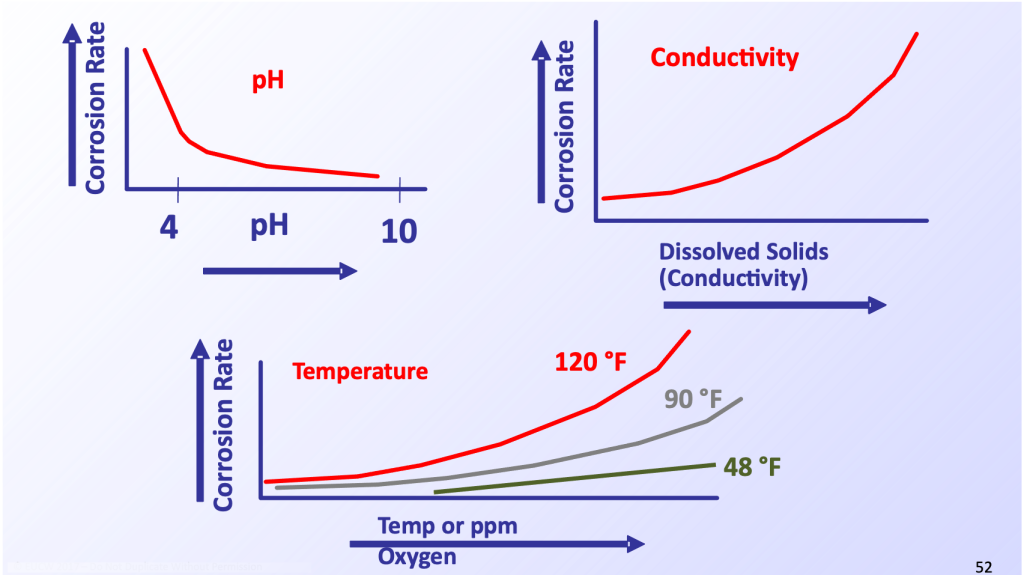
Corrosion is an electrochemical process, and as the dissolved solids concentration increases, so does the corresponding conductivity and corrosion rate. For some metals, and most notably carbon steel, the corrosion rate decreases with rising pH (except at very high pH where it increases again). The corrosion rate also usually increases with temperature and dissolved oxygen concentration. A mitigating factor at times when these two are in combination is that oxygen solubility decreases with increasing temperature. A dramatic example of this influence occurs in a specialized corrosion mechanism that this author has reported upon previously.5
Looking Forward to Part 2 and Beyond
With this background information in place, future installments will delve into a number of the most important corrosion mechanisms, including:
- General corrosion
- Localized corrosion mechanisms
- Pitting
- Crevice corrosion
- Fatigue
- Stress corrosion cracking, corrosion fatigue, and intergranular corrosion
- Galvanic corrosion
- Erosion corrosion
- Microbiologically influenced corrosion
Commonly, metal components for industrial and infrastructure applications are designed to provide a long lifetime when subject to just general corrosion. We will primarily focus on the localized mechanisms, some of which have been known to cause failures in months or even weeks in extreme cases.
References
- W.D. Callister, Jr., Materials Science and Engineering: An Introduction, 3rd Ed., John Wiley & Sons, 1994.
- P.R. Roberge, ed., Corrosion Basics: An Introduction, NACE International, Houston, Texas, 2018. (NACE is now part of the Association for Materials Properties and Performance, AMPP, still based in Houston.)
- Kitto, J.B., and Stultz, S.C., eds., Steam; its generation and use, Edition 41, Babcock & Wilcox, Barberton, Ohio, 2005.
- Post, R., Buecker, B., and Shulder, S., “Power Plant Cooling Water Fundamentals”; pre-workshop seminar for the 37th Annual Electric Utility Chemistry Workshop, June 6, 2017, Champaign, Illinois.
- B. Buecker, “HRSG Steam Generation Issues: Reemphasizing the Importance of FAC Corrosion Control, Parts 1-4” Power Engineering, September/October 2022. (www.power-eng.com)

Brad Buecker
Brad Buecker currently serves as Senior Technical Consultant with SAMCO Technologies. He is also the owner of Buecker & Associates, LLC, which provides independent technical writing/marketing services. Buecker has many years of experience in or supporting the power industry, much of it in steam generation chemistry, water treatment, air quality control, and results engineering positions with City Water, Light & Power (Springfield, Illinois) and Kansas City Power & Light Company's (now Evergy) La Cygne, Kansas, station. Additionally, his background includes eleven years with two engineering firms, Burns & McDonnell and Kiewit, and he spent two years as acting water/wastewater supervisor at a chemical plant. Buecker has a B.S. in chemistry from Iowa State University with additional course work in fluid mechanics, energy and materials balances, and advanced inorganic chemistry. He has authored or co-authored over 300 articles for various technical trade magazines, and he has written three books on power plant chemistry and air pollution control. He is a member of the ACS, AIChE, AIST, ASME, AWT, CTI, and he is active with Power-Gen International, the Electric Utility & Cogeneration Chemistry Workshop, and the International Water Conference. He can be reached at bueckerb@samcotech.com and beakertoo@aol.com.
Related Articles

The Importance of Equipment Fit, Tolerance, and Clearance

Debunking the Myths Around Bearings

Use the Right Grease for the Job

Pump and Driver Alignment 14-3

Setting Techniques for Tapered Roller Bearings

Should Jack Screws be tight or backed off?


