Major Corrosion Issues in Industrial Water Systems – Part 2: Microbiological Fouling
Brad Buecker, SAMCO Technologies
Posted 4/23/2026
Part 1 of this series outlined fundamental corrosion principles, including electrochemical basics. Equipment and infrastructure are designed with tolerance for general corrosion to maintain long-term integrity. However, many corrosion mechanisms are localized, in which seemingly slight metal loss, when focused in a small area, may cause rapid failures. The remaining parts of this series will focus on important localized corrosion mechanisms, with discussion about preventive measures that encompass such issues as corrosion control programs and materials selection. This installment examines localized corrosion induced by microbiological fouling.
Revisiting the Microbes
I covered important aspects of cooling water microbiological fouling in Part 2 of a previous series for Maintenance World,1 but the issue is so important that further discussion is appropriate. Microbial fouling can cause the most prompt and intense damage in cooling systems.
Microorganisms are everywhere, and they enter cooling systems from makeup water supplies and air flowing through cooling towers. Bacteria are the primary microbes of concern in cooling water networks and heat exchangers, while algae and fungi may be problematic on and in cooling towers. Free-floating bacteria are usually not troublesome on their own (unless pathological organisms are present), but if the bacteria are allowed to settle and form sessile colonies, the situation changes radically. When bacteria attach to surfaces, some organisms immediately begin to generate a protective, polysaccharide (slime) layer. The slime collects other microorganisms and suspended solids, and deposits can rapidly accumulate as shown in Figure 1.

Corrosion issues may be even more serious. Deposits in general can establish corrosion cells where the area beneath the deposit becomes anodic to bare metal and begins to corrode. (See Part 1 of this series for a discussion of anodes and cathodes.) The presence of small anodes in a large cathodic field generates pitting and potential through-wall penetrations in a short time frame. However, oxygen depletion corrosion is only part of the problem with microbiological fouling. Numerous organisms accumulate within a microbial colony.
Bacterial species gradually change within a colony according to depth. Aerobic bacteria that use oxygen for metabolism thrive near the surface. Facultative bacteria that can use oxygen for energy, but more often other oxygen containing compounds, are at mid-levels. Finally, and a key to this discussion, is the potential presence of anaerobic sulfate-reducing bacteria (SRB) at the lower levels of the colony and at the metal surface. SRBs utilize sulfate ions (SO42-) for respiration and release metabolic byproducts, in particular hydrogen sulfide (H2S), that are very destructive to many metals. Attack by such microbial byproducts generates what is known as microbiologically induced corrosion (MIC).

The following two case histories, one discussing MIC and the other outlining sulfide attack from a non-microbiological source, indicate the destructive capabilities of these compounds.
Case Histories
#1 – I directly witnessed a MIC case where, during a month-long scheduled maintenance outage, cooling water was allowed to remain standing in the 15,000 tubes of a power plant condenser. Upon startup, plant chemists immediately discovered excessive condensate contamination, which inspection revealed to be the result of thousands of pinhole leaks in the 304L stainless steel tubes. An entire tube replacement, at huge cost, was necessary. This is but one example of the importance of proper layup procedures for all water/steam touched circuits of steam generators. Look for a later series in Maintenance World that discusses layup issues.
#2 – This example illustrates the extreme corrosion potential of sulfides. A contractor replaced the aging Admiralty brass (70% Cu, 29% Zn, 1% Sn) tubes in a condenser with (usually) more durable 90-10 copper-nickel alloy. Within 18 months, numerous through-wall penetrations appeared in the new condenser tubes.

Subsequent investigation revealed that the material supplier had used a sulfide-containing lubricant during fabrication but did not completely remove the lubricant before shipping the product. The sulfides attacked the tubes in many locations. As in the previous example, this condenser required a complete tube replacement.
From a health standpoint, heavy microbiological deposits provide a habitat for Legionella pneumophila bacteria, which for decades have caused illnesses and deaths around the world. Space limitations prevent further discussion here, but additional information is available from the Cooling Technology Institute (www.CTI.org) and other organizations.
Microbial Preventive Measures
Reference 1 provided important discussion on microbial control measures, but some of the most important concepts bear repetition here.
For the large majority of recirculating and once-through cooling systems, oxidizing biocides are the backbone of microbial control programs, as this chemistry usually represents the most cost-effective method for maintaining system cleanliness. Near the turn of the 20th century, chlorine gas became the standard for drinking water and then cooling water applications for many years. One-ton cylinders were a common method of storage and supply. When chlorine is added to water the following reaction occurs:

HOCl, hypochlorous acid, is the killing agent, and it functions by penetrating cell walls and oxidizing internal cell components. Safety is obviously a major issue with handling chlorine cylinders and feeding chlorine gas, and years ago many industrial facilities switched to liquid sodium hypochlorite (NaOCl, aka bleach), with a common active chlorine concentration of 12.5%. The change to bleach allowed for use of metering pumps to feed the chemical, but a problem that many operators encountered was pump binding due to bleach’s tendency to vaporize in piping locations around the pump. Pump designs have evolved to prevent or relieve vapor lock.
Standard bleach treatment has limitations, especially in the modern cooling systems where scale/corrosion control programs operate at a moderately basic pH at or near 8. The efficacy and killing power of chlorine significantly decline with rising pH, per the equilibrium nature of HOCl in water, as shown below and in Figure 5.

OCl– is a much weaker biocide than HOCl, probably because the charge on the OCl– ion does not allow it to effectively penetrate cell walls.
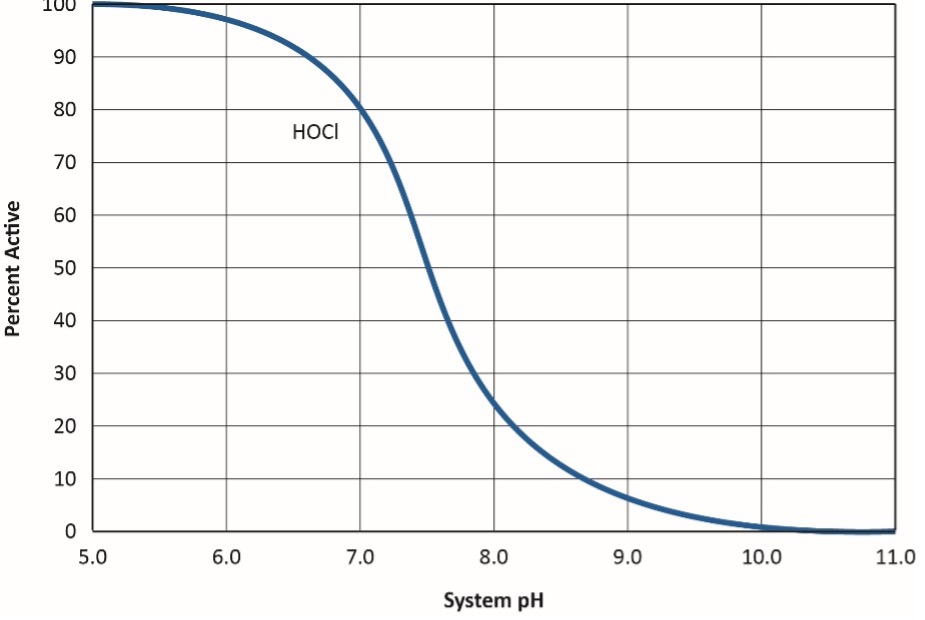
Figure 5. HOCl dissociation as a function of pH.2
The following sections outline alternative treatment chemistries, some of which enhance bleach potency, and some which are distinct compounds.
On-Line Hypochlorite Generation
One alternative to stored bleach is on-site hypochlorite generation.

Figure 6. An on-site hypochlorite generator. Source: https://morrowwater.com/miox/
The generator produces a mixed oxidant of hypochlorite and peroxide, from three common consumables: water, salt, and electricity. The process requires no storage of hazardous chemicals.
Mineral Oxychloride (MOCL)
The author was introduced to the new “MOCL” technology at the recent POWERGEN26/Electric Utility & Cogeneration Chemistry Workshop. The core chemical is bleach, but the process utilizes a proprietary catalyst that produces a variety of oxidizing compounds including hydroperoxyl (HO2–) and superoxide (O2–) ions, and others.3 The combined oxidizing power of these compounds is significantly higher than bleach or chlorine.
Bromine Chemistry
A popular answer to the bleach-pH difficulty mentioned above has been bromine chemistry, where a chlorine oxidizer (bleach is the common choice) and sodium bromide (NaBr) are blended in a slipstream and injected into the cooling water. The reaction produces hypobromous acid (HOBr), which has similar killing powers to HOCl but is more effective at higher pH. An eductor system with which the author worked for several years prepared HOBr by combining bleach with sodium bromide (NaBr) in a water side stream, then injected into main cooling water circuit.

An issue with the halogens, and especially chlorine, is their reactivity towards organic and nitrogen compounds, particularly ammonia. These reactions represent a halogen “demand” that reduces the effective biocide concentration. The reactions can also produce halogenated organics that may be regulated in the plant’s discharge permit.
Halogen Stabilizers
Several chemical compounds are available to stabilize chlorine and bromine and then release the oxidizers gradually, and where they are most needed. Stabilized halogens typically exhibit a lower oxidizing power than the parent halogen, but this reduced oxidizing power offers benefits with respect to microbial control in that it reduces undesirable reactions with the protective slime. Three classes of stabilizers dominate the market: sulfamate, dimethylhydantoin, and isocyanurates.
A supplemental treatment technique is feed of a bio-surfactant prior to the oxidizer. These molecules typically consist of a non-polar, hydrophobic carbon chain with a charged molecule at one end. The hydrophobic portion penetrates the organic slime matrix, with the active group being attracted to water. Surfactants/penetrants can loosen the slime layer allowing better penetration by the chemical agent. Most effective is to feed the surfactant perhaps a half-hour or so before the biocide to allow better penetration of the killing agent.
Chlorine Dioxide
Chlorine dioxide (ClO2) is a gas at room temperature that is stable and soluble in water to a maximum concentration of approximately 3,000 ppm. It must be prepared on site via the reaction of either sodium chlorite (NaClO2) or sodium chlorate (NaClO3) with an additional oxidizing agent under acidic conditions. Chlorine dioxide is more expensive than the halogens, but modern production techniques have lowered the cost.
Because chlorine dioxide exists as a gas in solution, it is easily stripped by aeration in cooling towers. ClO2 should be introduced below the surface of receiving waters to minimize losses. Handling of the chemical precursors for chlorine dioxide, which may include sulfuric acid, require attention to safety, although modern ClO2 generators are typically designed with safety in mind. Strict adherence to operational guidelines is important.
Chlorine dioxide is a selective oxidizer. For example, it does not react with ammonia and some organics and thus may be a good choice in waters containing significant concentrations of these “chlorine-demand” compounds.
Monochloramine
Chloramines have served for microbial control in water systems for over a century. In water containing ammonia, continued chlorine feed will produce a series of chloramines, starting from monochloramine (NH2Cl), then dichloramine (NHCl2), and finally nitrogen trichloride (NCl3). The solution reaches “breakpoint” once all ammonia has been consumed, upon which free chlorine appears. Monochloramine is the compound of interest for modern biofouling control, and technologies are now available to produce a pristine stream of NH2Cl for this purpose. When compared with sodium hypochlorite, monochloramine is less reactive but almost equivalently toxic. The reduced reactivity allows it to penetrate biofilms and attack underlying organisms. However, monochloramine generally needs a longer contact time than hypochlorite to achieve the desired microbial destruction.
Oxidizer Monitoring
Regular analysis of oxidizing biocide residual is extremely important to ensure consistent and proper dosages.
Prior to development of on-line monitoring methods, the standard procedure for halogen measurement was the DPD method by grab sampling. DPD is the acronym for N,N-diethyl-p-phenylenediamine. It reacts with oxidants in the water to form a colored complex, whose intensity is directly proportional to the amount of oxidant present. Different reagents are available for free and total halogen determinations.
Mature, on-line oxidizing biocide measurement is available. A standard industry instrument is shown below.

The analyzer extracts a sample, and per the DPD chemistry mentioned above, injects the chemical and a buffer solution. Following the proper reaction time, the instrument displays the oxidizer concentration, flushes the spent sample, and extracts a new sample. Thus, the analyzer can provide round-the-clock analyses, separated only by the time needed for reagent reaction. On-line monitoring can be of great benefit in promptly detecting biocide feed system malfunctions or water chemistry changes that increase biocide demand. Similar to grab-sample DPD analyses, either a free- or total-residual reagent is available for the analyzer.
Cooling waters typically contain numerous oxidizing or reducing compounds. Oxidation-reduction potential (ORP) measurement is an analytical technique that, as its name implies, provides data regarding the oxidizing or reducing power of the solution. The oxidizing potential of the oxidizing biocides usually dwarfs other species. ORP can serve as a good supplement to chlorine monitoring.
Non-Oxidizing Biocides
While oxidizing chemicals normally serve as the foundation of cooling water biocide programs, microorganisms can develop partial immunity. And, as has been noted, the slime produced by bacterial colonies also protects organisms. Feeding a non-oxidizing biocide on a periodic basis, e.g., once or twice per week for an hour or so, can help control microbial growth. Non-oxidizers typically penetrate cell walls to then react with compounds within the cell that are necessary for life. Programs can be tailored to address the most troublesome organisms in a system, whether they are bacteria, algae, or fungi.
On another note, and what could be considered Case History #3 of this article, the author, and other colleagues, have utilized non-oxidizing biocides for protection of reverse osmosis (RO) membranes from microbiological fouling. The most common RO membrane material, polyamide, contains nitrogen atoms that are readily attacked by chlorine. For makeup systems with chlorine or bleach raw water treatment, the halogen must be removed upstream of the RO unit. However, some microbes can go into hibernation until the chlorine disappears and then spring to life. I have seen severe fouling of membranes from this phenomenon. Periodic feed of a non-oxidizer, perhaps for an hour every two days, has proven effective in some applications.
Note: Plant personnel must receive approval from the proper regulatory authorities before making a change, or even testing, chemicals that are not currently authorized in the existing plant discharge permit(s).
Looking Forward to Part 3
Part 3 will continue the discussion of pitting attack, but from non-microbiological processes. Discussion will include important items of how material selection can influence pitting, and most notably, that stainless steels are not always the cure-all for corrosion that some people believe.
Disclaimer
This article offers general information and should not serve as a design specification. Every project has unique aspects that must be individually evaluated by experts from reputable water treatment firms.
References
- Buecker, B., “Protecting Cooling Water Systems – Part 2”; Maintenance World, July 2024.
- Post, R., Buecker, B., and Shulder, S., “Power Plant Cooling Water Fundamentals”; pre-workshop seminar to the 37th Annual Electric Utility Chemistry Workshop, June 6-8, 2017, Champaign, Illinois.
- Ordóñez, J.A., Oeschsner, S. and Rose, D., “Oxi-Plus: Advancing Microbial Control in Power Plant Cooling Water Systems”; presentation at the 43rd Annual Electric Utility & Cogeneration Chemistry Workshop, January 19, 2026, San Antonio, Texas. The EUCCW is now co-located with the annual POWERGEN conference.

Brad Buecker
Brad Buecker currently serves as Senior Technical Consultant with SAMCO Technologies. He is also the owner of Buecker & Associates, LLC, which provides independent technical writing/marketing services. Buecker has many years of experience in or supporting the power industry, much of it in steam generation chemistry, water treatment, air quality control, and results engineering positions with City Water, Light & Power (Springfield, Illinois) and Kansas City Power & Light Company's (now Evergy) La Cygne, Kansas, station. Additionally, his background includes eleven years with two engineering firms, Burns & McDonnell and Kiewit, and he spent two years as acting water/wastewater supervisor at a chemical plant. Buecker has a B.S. in chemistry from Iowa State University with additional course work in fluid mechanics, energy and materials balances, and advanced inorganic chemistry. He has authored or co-authored over 300 articles for various technical trade magazines, and he has written three books on power plant chemistry and air pollution control. He is a member of the ACS, AIChE, AIST, ASME, AWT, CTI, and he is active with Power-Gen International, the Electric Utility & Cogeneration Chemistry Workshop, and the International Water Conference. He can be reached at bueckerb@samcotech.com and beakertoo@aol.com.
Related Articles

Chain Drive Design Recommendations
The API Gland

Comparing the Value of Single- and Double-Layer Insulation on Boiler Walls

The Importance of Minimizing Corrosion Product Transport to Steam Generators

Best Practices for Compressor Storage

High-efficiency SynRM motors, a game changer in potentially explosive environments


