Root Cause Steam Generator Protection: Feeding the Proper Chemicals – Part 3
Brad Buecker, SAMCO Technologies and Buecker & Associates, LLC
Posted 8/19/2025
In a previous Maintenance World article, the author outlined the importance of reliable makeup water production to protect steam generators from deposition and corrosion. However, even with consistent high-purity makeup, boiler systems require chemical treatment at strategic locations to further inhibit corrosion and scale formation in the high-temperature environment. This installment outlines fundamental steam generator treatment concepts and common chemical feed locations for conventional, high-pressure power generation units. The discussion provides foundational information for a future article(s) outlining the nuances of water/steam chemistry treatment for modern combined cycle heat recovery steam generators (HRSGs) and lower-pressure industrial boilers, where physical conditions may be less severe but possibilities for impurity ingress are often greater.
Note: Following this series, we will examine chemical feed issues for cooling water systems.
A Core Aspect, pH Control
Carbon steel is the typical material of construction for most boilers and boiler feedwater piping. (In high-pressure utility units, higher alloy steels are needed for high-temperature superheaters/reheaters, and for turbines.) A key carbon steel corrosion control requirement is maintaining pH within a mildly alkaline range (~9-10) to minimize general corrosion. Similar reasoning applies, but with some modification, for those dwindling number of power plant units that have feedwater heaters with copper alloy tubes. Either way, pH control is a primary factor for treatment selection.
Utility Steam Generator Concepts
Many important boiler chemistry improvements came during the evolution of utility boilers in the middle of the last century. An important concept to remember for dedicated power units is that, within reasonable limits, the water/steam network is a closed system, where the steam, after expending its latent heat in the turbine, is condensed and returned to the boiler. Thus, the fluid continually recirculates, with minor losses such as blowdown and deaerator venting taken care of by the makeup system. Figure 1 outlines the common water/steam path of a large drum-type, coal-fired steam generator.

The list below offers a brief summation of important water/steam chemistry issues of which plant personnel became accustomed during the 20th century.
- The steam condenser is always a potential source of cooling water in-leakage to the condensate unless the unit has an air-cooled condenser. (Significant air in-leakage to the condensate and particulate iron formation are of concern for ACC-equipped power plants.) Major or chronic minor condenser tube leaks can be quite problematic.
- Makeup water is typically introduced to the network on the process side of the condenser. A makeup system upset offers another avenue for contaminant ingress to condensate. These events are rare (but not impossible) in steam generators having modern makeup water treatment technology.
- Condensate-feedwater corrosion control is important not only to protect the metal but to minimize formation of metal oxides that can precipitate on boiler tubes and establish sites for under-deposit corrosion (UDC).
- Boiler water treatment programs, and especially those utilizing phosphates, have gone through a somewhat complex evolution per the discovery over time of several different corrosion mechanisms.
- Boiler water chemistry guidelines are designed to protect boilers from corrosion but also to minimize carryover of impurities to superheater, reheater, and turbine components. These compounds may include sodium hydroxide, chloride and sulfate salts, siliceous compounds, and (becoming much less common) copper.
So, let’s look at a bit of history regarding how chemical treatment methodologies for power units emerged and evolved. We will divide the discussion between boiler water and boiler feedwater. People sometimes have trouble grasping that the feedwater system is not directly part of the boiler.
Boiler Water Chemistry Control
In the early days of power boiler development, makeup water treatment methods were not firmly established. Operators learned that the accumulation of dissolved ions in the boiler water, and especially the hardness ions calcium and magnesium, would induce scaling. The most common reaction was calcium carbonate (CaCO3) precipitation:


Compounding the issue is that CaCO3 is inversely soluble, meaning that the reaction is driven to the right with increasing temperature:

This effect is easily observable in non-industrial settings, for example in faucets, showerheads, etc., of home plumbing systems, where heated water lowers the CaCO3 solubility, such that hot water lines and fittings accumulate scale.
Other mineral deposits that still appear in industrial heat exchangers include (but are definitely not limited to):

Early treatment methods to combat deposition had operators dumping potato peels or sawdust into the boiler, as some of the organic compounds, i.e., tannins and lignin, in these natural materials sequester hardness. Of course, this approach was obviously not very scientific or practical. The development of ion exchange resins for sodium softening and then full demineralization greatly improved makeup water quality. However, it became apparent that internal boiler water treatment was still necessary to protect units – whose size, pressure, and complexity was growing – from both deposition and corrosion. In the 1930s, tri-sodium phosphate (Na3PO4) emerged as a primary treatment chemical. For starters, TSP generates alkalinity to minimize general corrosion.

Of greater importance, and especially in the early days when makeup water treatment was “iffy”, is that TSP and the alkalinity it produces would react with calcium and magnesium to form soft sludges, which can be removed via boiler blowdown. The alkalinity also neutralizes the effects of other impurities such as chloride and sulfate that can concentrate under tube deposits and generate acids, e.g., HCl. These acids will, of course, directly corrode boiler metal, but their reaction with iron generates hydrogen, which in turn may lead to hydrogen damage. In this mechanism, atomic hydrogen penetrates into the metal and combines with carbon atoms in the steel to generate methane (CH4). The voluminous methane molecules induce cracking and tube failure with little metal loss.

Guidelines for early power boilers called for phosphate concentrations in a range of up to 20-40 parts-per-million (ppm). However, as boilers increased in size and pressure/temperature, caustic gouging and embrittlement (the opposite conditions of hydrogen damage) became problematic due to the high phosphate dosage.
A further complication with phosphate chemistry is the relationship of TSP solubility with temperature.

The diagram clearly illustrates that TSP solubility rises with temperature to approximately 300o F, but then dramatically decreases thereafter. At the typical boiler water temperatures (600° F or greater) of utility steam generators, sodium phosphates are only slightly soluble and most of the chemical precipitates on boiler internals. This phenomenon, known as hideout, leaves little phosphate in solution to combat impurity ingress.
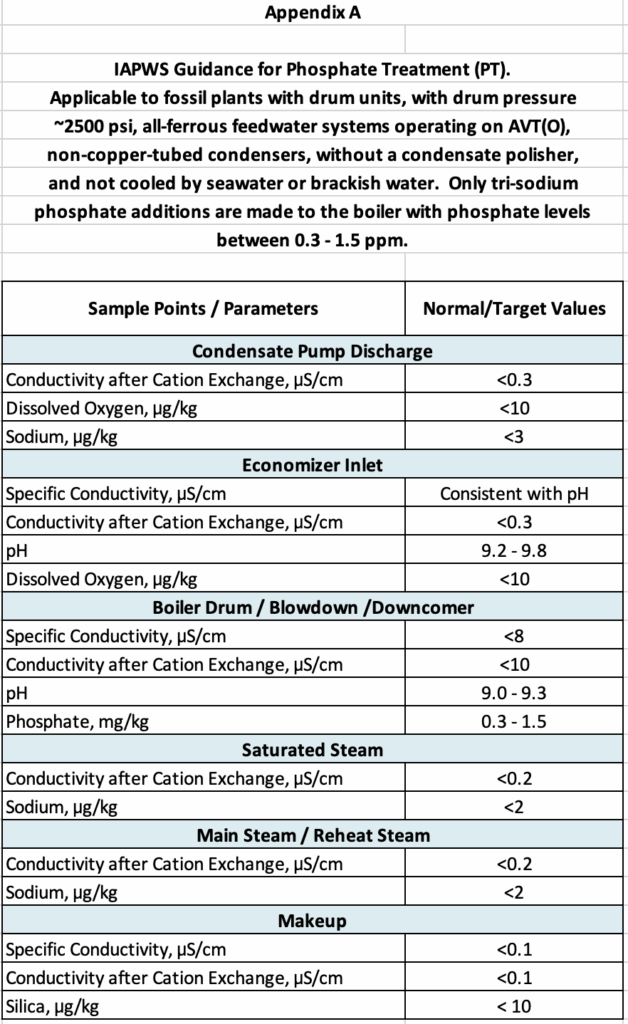
As researchers began to grasp these issues, a variety of phosphate treatment programs evolved, with names such as coordinated phosphate, congruent phosphate, and equilibrium phosphate. The first two included blends of TSP with disodium phosphate (Na2HPO4) and maybe even a bit of monosodium phosphate (NaH2PO4). The fine points (and deficiencies) of these chemistry regimes are too intricate for this article, but a more detailed discussion is available in Reference 3. A key concept is that TSP is the only phosphate species now recommended for high-pressure units, with PO4 concentrations maintained within a 0.3-1.5 mg/kg (mg/kg is equivalent to ppm) range to minimize hideout but provide initial protection against a condenser tube leak. The recommended feed point/configuration is a moderate diameter, e.g., 1-inch i.d., perforated pipe that extends along the length of the steam drum and below the water level.
A phosphate alternative (to eliminate hideout) is caustic (NaOH), but caustic treatment requires very careful control, per the afore-mentioned caustic gouging issues, and also because any excess caustic carryover to steam can cause prompt and serious stress corrosion cracking (SCC) of high alloy steels in superheater and reheaters, and especially turbines. This author would be reluctant to recommend caustic treatment unless the plant has an experienced chemistry staff and has the correct online instrumentation to continuously monitor steam and boiler water chemistry.
Note: Appendix A at the conclusion of this article comes from Reference 3 and outlines the general chemistry recommendations for a 2,500-psi unit on TSP treatment (and AVT(O) feedwater treatment, which is outlined in the next section). This guideline, as with any other, should never be considered a “one size fits all.” Every unit must be evaluated individually, with modification of operating limits as dictated by analytical data and unit performance.
Feedwater Treatment
The piping network from the condenser to the steam drum inlet constitutes the condensate/feedwater circuit of the steam generator. As noted, potential methods of impurity ingress are condenser tube leaks or, less likely, makeup system upsets. Online analytical instrumentation is important to alert operators and technical personnel to upset conditions. For routine operation, chemical feed is geared towards corrosion prevention to both protect the metal components and minimize transport of corrosion products to the boiler. When the author began his power industry career at a coal-fired plant in 1981, the common feedwater treatment program was known as all-volatile treatment reducing (AVT(R)). The standard method for feedwater pH control was, and generally still is, ammonia (NH3) feed.

NH3 is a volatile alkalizing agent, which is required because solid alkalis such as TSP or caustic are taboo for feedwater treatment, as they would be directly introduced to boiler steam via the attemperator system. A common ammonia injection point was once the deaerator storage tank/boiler feed pump suction. However, per the realization that the entire condensate/feedwater system would benefit from consistent pH control, feed at the condensate pump discharge became popular.
The influence of pH on carbon steel corrosion is illustrated in a well-known chart from the 1970s.

Evident is that the corrosion rate decreases with increasing pH. However, the equilibrium reaction of ammonia with water places an upper limit on effective concentration, as is clearly shown in the following table. We will revisit this issue when examining HRSG feedwater treatment in the next installment.
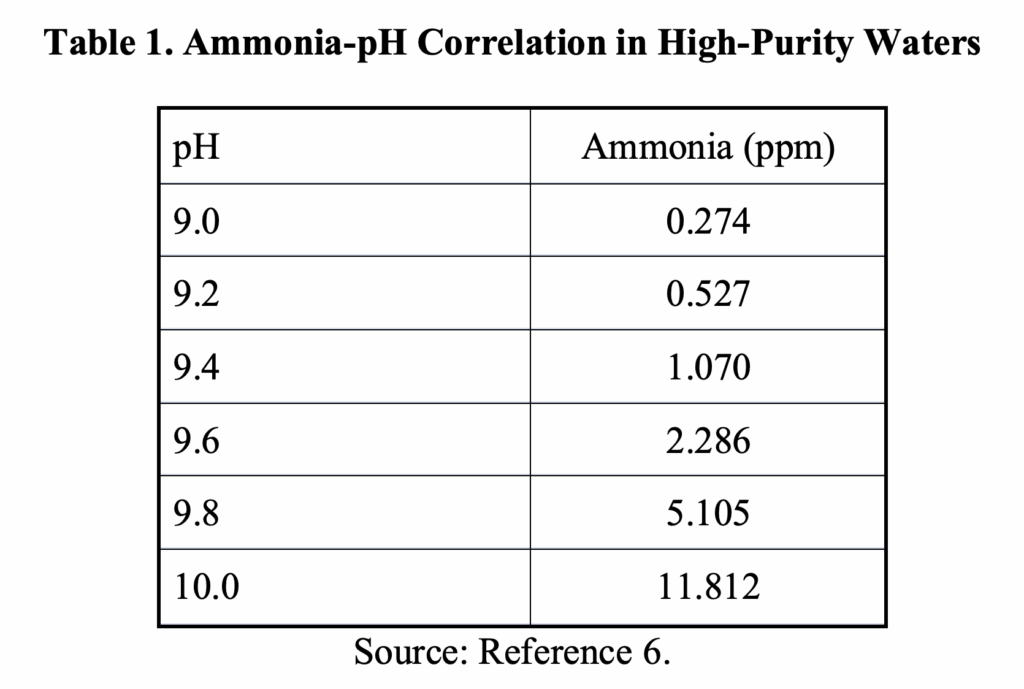
A mindset in the development of AVT(R) was that any dissolved oxygen (D.O.) in feedwater systems was deleterious and would cause extensive corrosion. This was certainly true for copper alloy feedwater heater tubes, where the combination of D.O. and ammonia for pH control can cause serious damage. A corresponding belief was that D.O. would also induce significant carbon steel corrosion, and indeed, uncontrolled (keep this word in mind) oxygen ingress can be quite corrosive to steel. Accordingly, virtually all U.S. power plant feedwater systems were equipped with a mechanical deaerator to reduce D.O. to approximately 7 parts-per-billion (ppb), with supplemental oxygen scavenger/reducing agent feed to further reduce D.O. concentrations to near zero. Aqueous hydrazine (N2H4) solutions were a popular oxygen scavenger initially, but concerns about the carcinogenic nature of the compound led to the development of alternatives.
The “zero oxygen” mindset received a transformative jolt in 1986, for “On December 9 of that year, an elbow in the condensate system ruptured at the Surry Nuclear Power Station [near Rushmere, Virginia.] The failure caused four fatalities and tens of millions of dollars in repair costs and lost revenues.” 6 The culprit was single-phase flow-accelerated corrosion (FAC) of carbon steel; a phenomenon that has since been discovered at many power plants, with additional fatalities from some FAC-induced failures. As the name implies, flow-accelerated corrosion occurs at flow changes or disturbances, including elbows in feedwater piping and economizers; feedwater heater drains; locations downstream of valves and reducing fittings; and attemperator piping. FAC induces gradual metal loss, which leads to sudden failure when the pipe wall can no longer withstand the pressure.

A primary influence for FAC is the presence of a reducing environment as produced by an oxygen scavenger. Also, as Figure 5 suggested, pH and temperature have strong influences; the latter especially within a range of around 200o to 400o F.
How has this problem been resolved? First, in the late 1960s/early 1970s chemists at supercritical power plants in Europe discovered that controlled oxygen injection into high-purity feedwater (and in units with no copper-alloy feedwater heater tubes) could provide substantial carbon steel corrosion protection by forming a different and more protective oxide layer than that formed from AVT(R) chemistry. The program became known as oxygenated treatment (OT), and it has been adopted at most supercritical units around the world. While OT is possible for drum units, scientists at EPRI developed a modified program known as all-volatile treatment oxidizing (AVT(O)), in which the typically small amount of oxygen that enters the condensate at the condenser is allowed to remain, combined with elimination of oxygen scavenger feed and perhaps also with deaerator vents closed during normal operation. In some cases, as we will explore in the next installment, supplemental oxygen feed may be needed to maintain sufficient feedwater D.O. concentrations.
A critical requirement for AVT(O) is high-purity feedwater (≤0.2 µS/cm cation conductivity). And, as with OT, AVT(O) cannot be utilized if the feedwater system has any copper alloys. Additional details are available in Reference 4. The author directly worked with EPRI to convert a large utility unit from AVT(R) to AVT(O) as a root cause preventive measure to minimize FAC and prevent any failures like that shown in Figure 6. During this project, I became personally aware of a FAC-induced failure in an attemperator feedwater line at a sister plant that caused two fatalities and seriously injured a third person. The key concept is that for most modern utility steam generators, AVT(R) is now strongly discouraged. Rather AVT(O) or OT should be the feedwater treatment method. Unfortunately, many power plant personnel around the globe have not grasped this idea and continue to employ AVT(R), with its potential to influence flow-accelerated corrosion.
Summary – Steam Generator Protection
This article presented fundamental ideas regarding power boiler chemical treatment programs and feed locations, as they first emerged in the early 20th century through evolution to the large units later in the century. The primary goal of these programs was root cause prevention of deposition and corrosion in utility units. From this foundation, we will examine the nuances of treatment methods for the modern multi-pressure heat recovery steam generators (HRSGs) of combined cycle power plants. Finally, we will look into treatment issues for industrial boilers, of which there are many thousands, where lower pressures allow some relaxation of impurity concentrations, but where multiple condensate returns may make the potential for impurity ingress much greater.
Disclaimer
This article offers general information about steam generator protection and should not serve as a design specification. Every project has unique aspects that must be individually evaluated during project design and subsequent operation. From those evaluations, comprehensive specifications can be developed and adjusted.
References
- Buecker, B., and Shulder, S., “Combined Cycle and Co-Generation Water/Steam Chemistry Control”; pre-workshop seminar for the Annual Electric Utility Chemistry Workshop, June 7, 2022, Champaign, Illinois.
- Buecker, B., and Shulder, S., “The Basics of Power Plant Cycle Chemistry”; pre-workshop seminar for the 27thAnnual Electric Utility Chemistry Workshop, May 2007, Champaign, Illinois.
- International Association for the Properties of Water and Steam, Technical Guidance Document: Phosphate and NaOH treatments for the steam-water circuits of drum boilers of fossil and combined cycle/HRSG power plants(2015). IAPWS technical guidance documents can be downloaded at no charge from www.iapws.org.
- International Association for the Properties of Water and Steam, Technical Guidance Document: Volatile treatments for the steam-water circuits of fossil and combined cycle/HRSG power plants (2015).
- Sturla, P., Proc., Fifth National Feedwater Conference, 1973, Prague, Czechoslovakia.
- Guidelines for Control of Flow-Accelerated Corrosion in Fossil and Combined Cycle Power Plants, EPRI Technical Report 3002011569, the Electric Power Research Institute, Palo Alto, California, 2017. This document is available to the industry as a free report because FAC is such an important safety issue.

Brad Buecker
Brad Buecker currently serves as Senior Technical Consultant with SAMCO Technologies. He is also the owner of Buecker & Associates, LLC, which provides independent technical writing/marketing services. Buecker has many years of experience in or supporting the power industry, much of it in steam generation chemistry, water treatment, air quality control, and results engineering positions with City Water, Light & Power (Springfield, Illinois) and Kansas City Power & Light Company's (now Evergy) La Cygne, Kansas, station. Additionally, his background includes eleven years with two engineering firms, Burns & McDonnell and Kiewit, and he spent two years as acting water/wastewater supervisor at a chemical plant. Buecker has a B.S. in chemistry from Iowa State University with additional course work in fluid mechanics, energy and materials balances, and advanced inorganic chemistry. He has authored or co-authored over 300 articles for various technical trade magazines, and he has written three books on power plant chemistry and air pollution control. He is a member of the ACS, AIChE, AIST, ASME, AWT, CTI, and he is active with Power-Gen International, the Electric Utility & Cogeneration Chemistry Workshop, and the International Water Conference. He can be reached at bueckerb@samcotech.com and beakertoo@aol.com.
Related Articles

Test for Pumping System Efficiency

Strive to Improve Your Gearbox Performance

Plant Numbering and Asset Identification

Increasing Pump Reliability and Life

The State of the Semiconductor Industry and Its Growth

Chain Drive Design Recommendations


